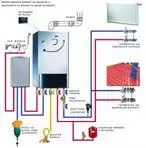
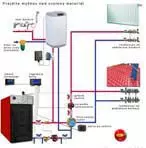
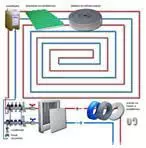
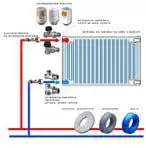
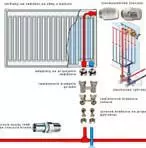
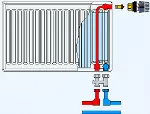
metal
A metal (or, rarely, a metal) is chemically a crystalline, electropositive substance with free valence electrons. These electrons are common to all atoms of the metal and can move freely in it. They are called free electrons. The property of metals to conduct an electric current through free electrons is called the electronic conductivity of metals.
Features
High electrical and thermal conductivity depends on the temperature of the metal. A rising temperature causes an increase in electrical resistance, a falling temperature improves conductivity. At temperatures close to 0 K, metal conducts electricity absolutely (without electrical resistance). This phenomenon is called superconductivity. In the solid state, metals have plasticity (malleability, formability), are able to reflect light and are opaque (light does not pass through them). These properties are related to the presence of free electrons.
In nature, metals occur mainly in compounds - ores. Some metals that are more difficult to form compounds can also be found in their pure form. Metals that easily form compounds, on the other hand, cannot exist in their pure form, they gradually corrode in the air (usually oxygen compounds, or a compound with a bond to free gases - sulfur, nitrogen...). Corrosion of metals is significantly promoted by water.
Toxic metals are metals that have toxic effects (they are poisonous to the human body), e.g. cadmium, mercury, lead, heavy metals.
Ferromagnetic metals are metals with magnetic properties (Co, Ni, Fe).
Materials that are composed of atoms (molecules) of several metals (or metals with non-metals while preserving the properties of the metal) are called alloys.